
Pourbaix diagrams are commonly given at room temperature, atmospheric pressure, and molar concentrations of 10 −6 and changing any of these parameters will yield a different diagram. Beside potential and pH, the equilibrium concentrations are also dependent upon, e.g., temperature, pressure, and concentration. 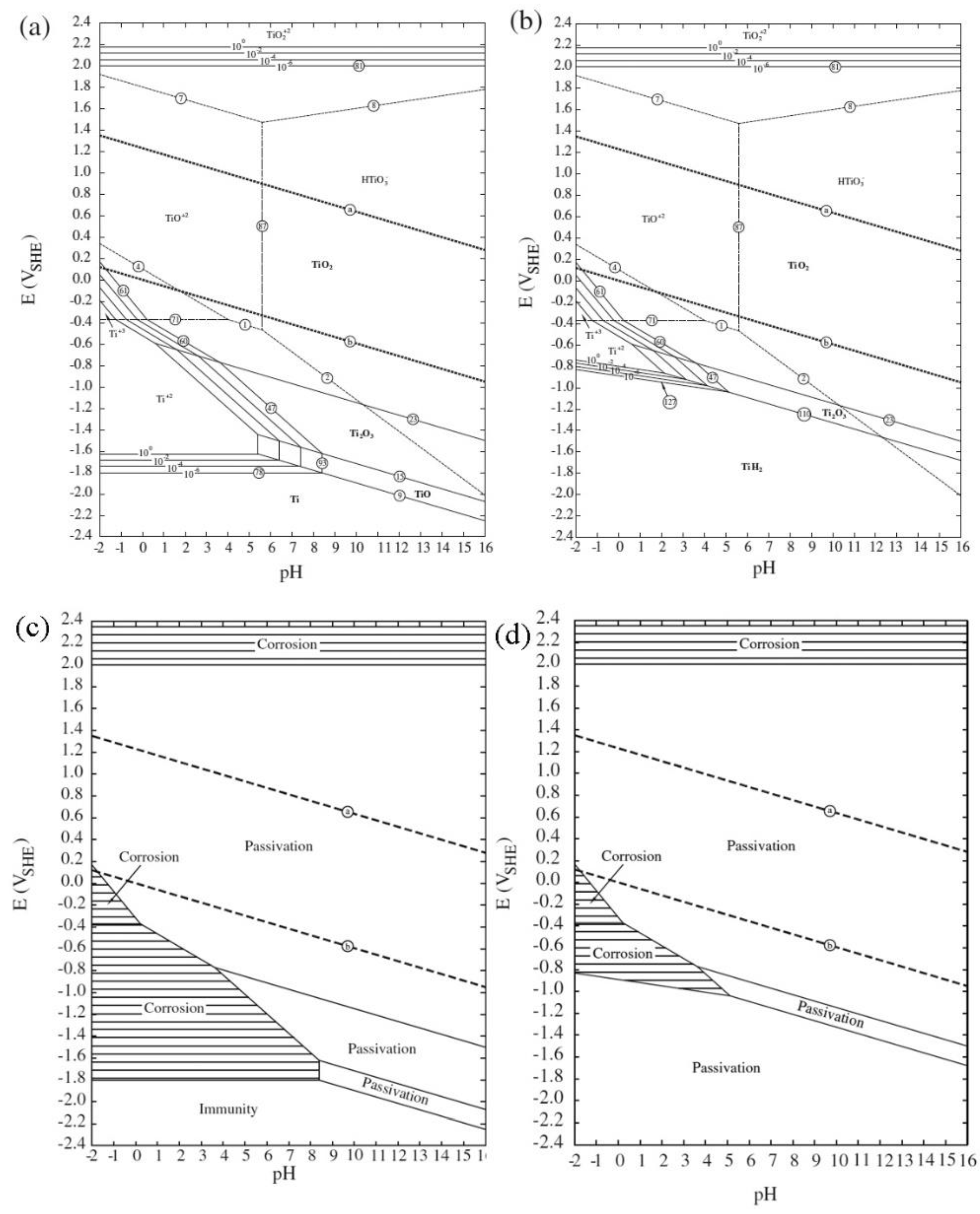
Similarly to phase diagrams, they do not allow for reaction rate or kinetic effects. As such a Pourbaix diagram can be read much like a standard phase diagram with a different set of axes. Boundaries (50 %/50 %) between the predominant chemical species (aqueous ions in solution, or solid phases) are represented by lines. In electrochemistry, and more generally in solution chemistry, a Pourbaix diagram, also known as a potential/pH diagram, E H–pH diagram or a pE/pH diagram, is a plot of possible thermodynamically stable phases ( i.e., at chemical equilibrium) of an aqueous electrochemical system. The Y axis corresponds to voltage potential.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
